Polio virus: vaccination ‘catch-up’ campaign rolled out…
[…]



ITSU has three verticals namely Program Implementation, Strategic Communication and Adverse Event Following Immunization. Currently, ITSU has three ongoing programs Routine Immunization, COVID-19 Vaccination and Urban Immunization which are being implemented in close partnership with MoHFW and State Health Departments.
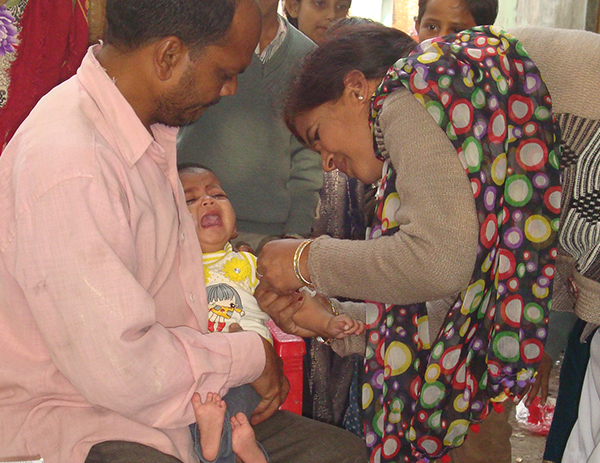





Being the technical arm of the Immunization division of the Ministry of Health & Family Welfare, Government of India, ITSU supports the government in formulating technical guidelines for new vaccine introduction, ramping up routine immunization coverage, finalizing standard operating procedures (SoPs) and other information, education and communication (IEC) and behavior change communication (BCC) materials.
Several best practices and successes were recorded during the course of ITSU’s work in scaling up immunization and new vaccines especially in far-flung areas, urban slums and amongst disadvantaged population across states and districts. COVID-19 vaccine roll-out also demonstrated resilience of communities and dedication of frontline workers.


Immunization Technical Support Unit (ITSU) provides techno-managerial support to Ministry of Health and Family Welfare (MoHFW) for scaling up the Universal Immunization Programme (UIP).
© 2022, Immunization Technical Support Unit, Ministry of Health and Family Welfare. | Designed by Media Solutions